Nanoparticles are of great scientific interest as they are effectively a bridge between bulk materials and atomic or molecular structures. A bulk material should have constant physical properties regardless of its size, but at the nanoscale this is often not the case. Size dependent properties are observed such as quantum confinement in semiconductor particles, surface plasmon resonance in some metal particles and superparamagnetism in magnetic materials.
The interesting and sometimes unexpected properties of nanoparticles are partly due to the aspects of the surface of the material dominating the properties in lieu of the bulk properties. The percentage of atoms at the surface of a material becomes significant as the size of that material approaches the nanoscale. For bulk materials larger than one micrometre the percentage of atoms at the surface is minuscule relative to the total number of atoms of the material.
Nanoparticles often have unexpected visible properties because they are small enough to scatter visible light rather than absorb it. For example gold nanoparticles appear deep red to black in solution.
The properties of materials change as their size approaches the nanoscale. For example, the bending of bulk copper (wire, ribbon, etc.) occurs with movement of copper atoms/clusters at about the 50 nm scale. Copper nanoparticles smaller than 50 nm are considered super hard materials that do not exhibit the same malleability and ductility as bulk copper. Carbon and MoS2 nanotubes are showing extremely high mechanical stabilities in comparison to bulk Carbon or MoS2 and are therefore of great scientific interest.
Suspensions of nanoparticles are possible because the interaction of the particle surface with the solvent is strong enough to overcome differences in density, which usually result in a material either sinking or floating in a liquid. But without further surface modification, nanoparticles tend to form agglomerates. To overcome this difficulties, tensed are used to stabilize the particles. In most cases small molecules are used to adsorb to the nanoparticle surface. But there are issues about using small molecule surfactants because there is a dynamic equilibrium of bound and unbound surfactants in solution. This makes it necessary to work with an excess of surfactant in solution so that micelles of pure surfactant are not avoidable.
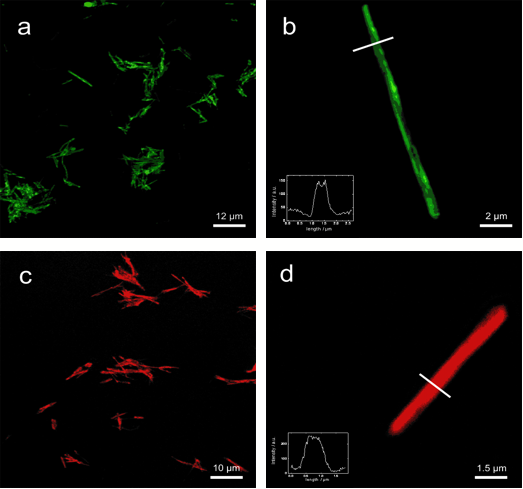
To overcome this problem, our group has focused on polymeric surfactants. These are made of block copolymers or statistic copolymers of anchor groups, soluble groups. In advance it is possible to connect dyes to the polymers to create unique visible properties.

surface modified TiO2 nanorods (confocal fluorescence microscope images)
Our group is using controlled radical polymerizations like RAFT, ATRP or NMP to create functional polymers with low polydispersities. With these techniques, block copolymers can be made and by designing the starting reagents of the polymerizations it is possible to create α,ω functional polymers. By the usage of reactive monomers (LINK REAKIVESTERCHEMIE) one can create very effective surfactants like shown in the diagram below.
Instead of a dye one can bound enzymes to the polymer that catalyses the deposition of gold nanoparticles for example.

SEM image of TiO2 nanorods with small gold particles at the surface
Nanotubes or nanorods are very interesting because of their anisotropy. The properties of these particles are not only different from bulk material but also depend on the direction of the particle. When those particles are well enough solubilized, they can form arrays of nanoparticles. These inorganic liquid crystalline structures are supposed to show very unique properties which depends on the orientation of the director.
